Physical activity and nutritional supplementation to reduce frailty in community- dwelling older adults, searching for evidence: a systematic review of randomized controlled trials
Laura Morciano,1 Gennaro Cerone,1 Francesco Cerutti,1 Francesca Di Gaspare,1 Claudia Alessandroni,1 Francesca Lucaroni,1 Cristina Ambrosone,1 Mario Messinese,1 Francesca Paradiso,1 Francesco Gilardi,1 Giuseppe Liotta,1 Sandro Mancinelli,1 Leonardo Palombi1
1 University of Rome Tor Vergata
Background
In the last decades, older people represent a quickly growing proportion of the world’s population. In 2015, 8.5 percent (617 million) of people worldwide were aged 65 and over. According to recent data, this is expected to grow to 17 percent (1.6 billion) by 2050.1 In developed countries, this percentage reaches higher levels, with a peak in Italy, where in 2016 elderly people accounted for the 22% of the resident population.2 Ageing population represents one of the most significant transformations of the twenty-first century, with implication for nearly all sectors of society, such as housing, transportation and social protection.3 Moreover, the increase in ageing represents many opportunities and several public health challenges to which the health systems needs to prepare for. Primary, a strong shift from hospital-based to community-based care is suggested to address the burden of multi-morbidity and its effects on adverse healthcare outcomes.4 In fact, due to physiological aging, the elderly reveal a reduction of psycho-physical efficiency that sometimes results in a feature of a greater vulnerability, also called frailty.5 Frailty in the elderly is defined as a clinical syndrome characterized by a decrease in energy reserve, strength, and performance, resulting in a cumulative decline in multiple physiological systems, leading to a state of greater vulnerability.6
There are several ways to approach to frailty.7 The most known of these was introduced by Fried and collaborators in the 2001, the “frailty phenotype”. It is based on the identification of frailty when 3 or more of these criteria are present: unintentional weight loss, self-reported exhaustion, weakness, slow walking speed, and low physical activity.8 A different model of frailty was proposed by Rockwood and colleagues.9 This approach defines frailty as a result of multiple interacting factors such as having difficulties in activities of daily living (ADL), psychological and social dimensions.10 The most recent approach to frailty was proposed in 2010 by Gobbens and colleagues, which defined frailty as a multidimensional syndrome.11 To date, a consensus on the exact definition of frailty is still lacking.4 Clinically, frailty is a potentially reversible syndrome12 that determines an increased risk of adverse health outcomes when exposed to a stressor.13 Indeed, frailty represent a major risk factor for negative health-related events in the elderly, including disability, falls, the need of long term care, hospitalization and death.14,15 Recent data suggest that the prevalence of frailty ranges between 4.0% to 17.0% (mean 9.9%) in the elderly, with higher percentage (13.6%) when psychosocial frailty was considered. This condition was increased in persons older than 80 and women had almost twice the risk of men (9.6% vs 5.2%) to be frail.16
Therefore, clinical identification of frailty may play an important role in its prevention.14 Due to its reversible nature, preventive strategies can be considered able to modify the natural history of this syndrome, with remarkable differences among community-dwelling and hospitalized frail individuals.17,18
The positive impact of physical exercise and nutritional interventions on frailty have long been investigated,19 with uncertain results; in fact, in recent literature, physical activity is considered one of the most effective intervention in frail older persons, while separate nutritional intervention doesn’t show noteworthy improvements. Recent data seem to associate type of dietary with a higher or lower risk of frailty. Despite the wide literature about this topic, a very few studies examined the reduction of frailty as primary outcome, and more often the population study doesn’t respond to an exact definition of frailty.
The aim of this study was to evaluate the most effective intervention in reducing the frailty status, considering both physical activity and nutritional intervention in community-dwelling older adults, for highlight the better strategy in reducing the prevalence of frail elderly.
Methods
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement was followed as a methodological guide.20
A systematic literature research was performed in November 2016 through PubMed and Cochrane Central Register of Controlled Trials, using the following keyword: frail elderly and frailty as MeSH terms, physical activity or nutritional, exercise or nutrition, training or protein, amino acids, supplementation in the title/abstract. The searching was restricted to clinical trials published in the last 10 years, written in English.
As showed in Table 1, reporting inclusion criteria, clinical trials were considered eligible if included both gender population of frail community-dwelling, 65 years old or more excepted for one study21 that included people from 61-89 years, self-dependent and without disabling diseases.
Inclusion criteria of randomized clinical trials
A specific definition of frailty was mandatory for eligibility, according to standardized criteria (eg, Fried’s criteria or Frailty Index).
Selected articles had to evaluate the effect of active physical exercise programs, nutritional supplementation, or both on frailty status. As secondary outcomes, selected studies had to evaluate the performance-based measures of physical function, in frail older adults. All the studies should have the presence of a control group and randomization in the assignment to each group (Table 1).
Studies were not included if anyone of the following participants’ characteristics was present: people living in a health structure or senior facility (hospitals, long-term care), patients with outcome-related comorbidities (e.g. diabetes, cardiovascular diseases), relevant acute recent event (as post-stroke, post fracture, etc.), severe cognitive impairment, single gender cohort.
The primary outcome was the evaluation of frailty status before and after the intervention. Secondary outcomes were the evaluation of other parameters such as disability, depressive symptoms, health-related quality of life, nutritional status, cognitive status, social networking, biochemical markers, self-reported health status.
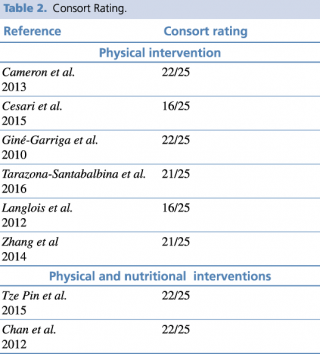
Methodological quality of all the selected studies was evaluated using the CONSORT (CONsolidated Standards of Reporting Trials) 2010 checklist for randomized trial.22,23 One point was assigned for each respected item of the checklist. Quality evaluation was independently achieved by four authors (C.A., G.C., F.C., F. DG.).
Four authors (C.A., G.C., F.C., and F. DG.) independently screened in pairs the search results, performed data extraction and quality evaluation. Any discrepancies were resolved by consensus or consulting with a fifth author (L.M.).
Results
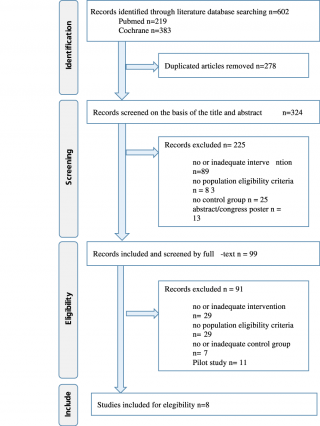
The two-database search identified 602 articles totally, and after duplicated removal, 324 were considered for selection about title and abstract. Through the reading of title and abstract, 225 articles were excluded, and 99 were retrieved for full-text assessment of eligibility. Eight studies met the inclusion criteria and were included in this review.
Details about selection workflow are shown in the Flowchart (Figure 1).
Eight studies satisfied eligibility criteria, for a sample of 1265 older people (64.6% women) with a mean age of 77.64 years. Two studies were conducted in Europe,24,25 one in Australia/Oceania,26 two in Asia27,28,29 and two in North of America.21,30 Follow-up measurements ranged from 3 to 12 months. Six selected articles were centered only on physical intervention, and two were based on both physical and nutritional intervention. No studies regarding only nutritional intervention were included.
Selected studies were of good quality, with a mean score of 20.25 out of 25, ranging from 16,21,30 moderate quality, to 22 points,24,26,28,29 excellent quality, as shown in Table 2. Assignment randomization methods were exhaustively described in six articles;24,25,26,27,28,29 due to the nature of interventions, none of the studies had blinded participants. Regarding the evaluation of outcomes, articles were divided in two groups per type of intervention: only physical intervention21,24,25,26,27,30 or both physical and nutritional intervention.28,29
A qualitative synthesis of the studies meeting the eligibility criteria or details about the data items are reported on Table 3.
Consort rating of selected articles
Intervention Characteristics
The intervention characteristics are summarized in Table 2. In all the selected studies an exhaustive description of intervention program was reported. Six studies21,24,25,26,27,30 were characterized only by physical interventions. In the study of Giné-Garriga et al.24 population was submitted to a functional circuit training program, focusing on improvement of balance and lower body strength. Cameron et al.26 conducted a multifactorial, interdisciplinary and individually tailored treatment intervention, targeted on better mobility and better balance, with a weight-bearing exercise program. Tarazona-Santalbina and collaborators25 performed a multicomponent exercise program focused on improving balance, proprioception, and strength, adding nutritional information on optimal dietary intake. Cesari et al.30 based their research on intervention including aerobic, strength, flexibility, and balance training. Langlois et al.21 performed a physical training sessions including a warm-up session, a strength training and an aerobic workout that was individually increased to reach a moderate to hard intensity, followed by a set of cool down exercises. Zhang et al.27 considered the whole-body vibration, which is a passive strong physical therapy, as the physical intervention, compared to a control group doing other kind of physical therapies and routine exercises.
Two studies26,27 investigated the effects of both physical intervention and nutritional program, combined and individually, compared to a control group. In the study of Ng et al.28 was performed a moderate physical exercise of 90 min/twice a week for 12 weeks in classes with a trainer, followed by 12 weeks of home-based exercises. Iron, folate, vitamin (B6, B12, D) and calcium supplement were administered daily for 24 weeks. Chan et al.29 based the principal intervention (exercise and nutritional) in a structured exercise course 3 times a week for 3 months. Each section lasted about 1 h. The research team also inquired about the subjects’ dietary compliance and responded to their dietary questions during the exercise sessions.
The interventions lasted 2 months in one study,27 3 months in three studies,21,24,29 6 months in two studies,25,28 12 months in the last two studies.26,30
The frequency of the interventions ranged from twice a week24,28 to 5 times/week25,27 for physical activity, while nutritional interventions were dispensed daily for all the time of the study.
Table three summarized in detail primary significant outcomes. A p value <0.05 or <0.001 were chosen as significance threshold level.
Outcomes
The primary outcome of this review was to investigate the potential reversibility power of a physical or combined physical and nutritional intervention program on frailty status.
Five of the included trials25,26,28,29,30 evaluated the frailty status as an outcome and all the studies showed a significant improvement on specific frailty scores in the intervention group compared to the control one. Among the studies which investigated the effect of only physical intervention, both Cameron et al.26 and Cesari et al.30 evidenced an improvement of Fried-CHS criteria (p<0.01 at 12 months and p=0.01 at 12 months, respectively). Even Tarazona-Santabalbina et al.25 evaluated the effect of a physical intervention alone, measuring Fried frailty criteria and Edmonton Frailty Scale at 24 weeks, showing a significant improvement of both frailty parameters (p<0.001), compared to the control group. Furthermore, Ng et al. and Ding-Cheng et al.,28,29 evaluated the effect of a combined physical and nutritional intervention on frailty status. Ding Cheng et al.29 showed a significant improvement of Fried-CHS score in a combined intervention group compared to the control (P=0.008 at 3 months); Ng et al.28 observed a significant improvement in Fried Frailty Criteria at 12 months in the combined physical and nutritional intervention group (p<0.01), moreover showing a less but still significant positive effect on frailty status (p<0.05) of an only nutritional supplementation strategy.
Other relevant outcomes were observed, such as mobility, muscle strength, balance, quality of life, cognitive status and biochemical markers.
Mobility outcomes were investigated in all the trials evaluating the only physical intervention. Five authors21,24,25,26,27 showed a statistically significant improvement in several mobility parameters: 6 minute walking test21 (p=0.03 at 12 weeks), short physical performance battery (SPPB)25,26 (p<0.001 at 12 months and p=0.007 at 24 weeks, respectively), lower extremity continuous summary performance score (LECSPS)26 (p<0.001 at 12 months), physical performance test (p<0.001) and Tinetti scales (p<0.001),25 time up and go and 30 second chair stand tests27 (both p<0.05 at 8 weeks). Gine-Garriga et al.24 evaluated a battery of several mobility tests, which almost all significantly improved at 36 weeks, such as stand up test (p=0.002), rapid gait test (p<0.001), normal speed (p=0.003), fast speed (p=0.001). In one of the trials evaluating a combined intervention,28 a gait speed test was significantly improved (p<0.05) in the group undergoing an only physical training. Two authors25,30 evaluated mobility indicators but they didn’t found significant improvements. No mobility outcome was evaluated in one combined intervention trial.29
Several muscle strength parameters were significantly improved in four trials24,27,28,29 after intervention programs, while in two studies21,30 intervention did not prove to have relevant effects. Ginè-Garriga et al.24 detected an efficacy of physical intervention on isometric dynamometry performance at 12 weeks (p<0.001); Li et al.27 measured the efficacy on knee extensor strength (p<0.05 at 8 weeks). Ng et al.28 showed a knee extensor strength improvement in both physical and combined intervention groups (p<0.01 at 12 weeks), while no significant improvement was observed in the only nutritional supplementation group. Ding-Cheng et al.29 evaluated the effect of combined intervention on dominant leg extension power, not showing any statistical improvement.
Balance scales were significantly improved in two articles24,27 in which a physical intervention was performed. Ginè-Garriga et al.24 evidenced at 36 weeks an improvement in tandem (p<0.001) and semitandem (p=0.021) tests, while single-leg test significantly improved (p<0.001) from 12 to 36 weeks. Li et al.27 demonstrated the efficacy of a whole-body vibration training session on frail elderly’s balance capacity at 8 weeks through an elliptic surface area walking (p<0.05). No balance scales were evaluated in the other included trials.
After his physical training program, Langlois et al.21 measured an improvement of an Achieve Personal Goals in 28 life domains as a quality of life indicator (p<0.05). Physical exercise was effective in improving Barthel index in two studies24,25 (p=0.001 at 36 weeks and p<0.001 at 24 weeks, respectively). Cameron et al.26 and Ng et al.28 evaluated a quality of life assessment with different scales with no statistically significant results at follow-up.
Anthropometric parameters, such as BMI, were measured as outcomes in three trials and did not show a significant benefit neither after a physical exercise program24,25 nor after a combined physical and nutritional intervention.29
Physical and nutritional combined intervention, in Chan et al.,29 showed to have significant effects on increasing in 25 (OH) vitamin D levels at 12 months (p=0.006); Tarazona-Santabalbina25 evidenced a relevant improvement of another biochemical marker such as the plasma protein carbonyls levels at 24 weeks (p=0.05), performing only a physical program.
Different cognitive status scales were investigated in three trials21,25,27 in which a significant better score was recorded after intervention, while in a combined intervention Chan et al.29 did not observed any significant improvement in MMSE at follow-up. Langlois et al.21 applied a cognitive domain survey which showed a significant score raising at 12 weeks (p<0.05); Tarazona-Santabalbina et al.25 detected a significant improvement (p=0.025) of MMSE at 24 weeks; Li et al.27 evaluated a Mental Health Component Score (MCS) which was significantly improvement at 8 weeks (p<0.05). Cameron et al.26 recorded no statistically significant changes on a depression scale values during the follow-up in the intervention group.
Discussion
This systematic review highlighted that both interventions, physical activity alone or physical and nutritional combined, have undoubted positive effects on frailty status. Moreover, in all the studies based on physical activity intervention, it was observed an important and statistically significant improvement of physical parameters considered as frailty criteria. Furthermore, in articles that considered both physical and nutritional interventions, the improvement comprises also some chemical biomarker. Other frailty-related parameters, such as cognitive, balance and lower extremities performance indicators, Activity Daily Living and other generic quality of life indicators have been improved in the study populations compared to the control groups. All the trial included in our paper were of good quality, but probably the strictly inclusion criteria could have reduced the number of studies included, with a scarce prevalence of nutritional clinical trials. Nevertheless, in the last years several systematic reviews were published among the most effective interventions in frailty, but none, to date, exploring the combined effect of physical activity and nutritional implementation. The only review that affords this topic is a scoping review published in January 2017, where differences with our work are remarkable.31
The effects of physical activity on frailty are more studied. A heterogeneity of definitions, tools and results is the biggest challenge that researchers encountered. In fact, most studies reported in the literature, differ in terms of included participants (frailty diagnostic tools), intervention strategies (type, number and combination of interventions) and intervention duration.32 As expected, also in our study physical exercise, with or without a nutritional intervention, improves frailty parameters, confirming the reversible nature of frailty syndrome and the importance of prevention or early intervention.
In some recent systematic reviews,33,34,35 the effects of exercise intervention in frail older people could promote an implement of physical functioning, but results showed great heterogeneity in terms of outcomes. De-Labra et al.35 concluded that multi-component exercise interventions, including some type of resistance training, could promote better performance in the global function capacity of frail older adults. Chou et al.34 showed a significant benefit of exercise in gait speed and in improving balance, but also great heterogeneity in results for the timed up and go test (TUG test) and performance in ADL. Giné-Garriga et al.33 found moderate evidence to support exercise training for improving gait speed and combined performance measures such as SPPB in agreement with other authors,34 but inconclusive findings about the effect of exercise training for improving functional mobility or balance, in contrast to other authors.34,36 In conclusion, the exercise training has some benefit in frail older people, but uncertainty still exists regarding which exercise characteristics (type, frequency, duration) are most effective. According to our results, it seems that physical activity intervention itself could improve the frailty status, regardless of the type of exercise. In our revision, most of the beneficial effects of the physical intervention were observed also over time (12 months), highlighting the importance of this kind of intervention in improving quality of life and reducing the institutionalization and/or hospitalization of frail older adults. In fact, one of the utmost important consequences of frailty is the onset of disability. Ramon Daniels et al.36 investigated how to prevent disability in frailty community-dwelling elderly through physical interventions. They were focused on disability outcome, where disability was defined as having trouble in performing activities in any domain of life. Exercise intervention seems to have a positive effect also on ADL and IADL in community-living people with a moderate physically disability.
Nutritional supplement doesn’t seem to modify the frailty status, but it should attend more frequently on the metabolic aspect, improving the hematologic pattern, and the concentration of certain nutrients used as nutrition markers. Noteworthy, nutritional status is an important predictor marker of frailty. As reported by Yannakoulia M et al., all the frailty criteria are affected by poor eating habits.37 They analyzed cross-sectional studies, prospective studies and intervention studies, assessed that not only a low level of energy intake is consistently associated with frailty, but also the daily distribution of dietary intake, the number of micronutrient deficiencies and the dietary patterns contribute to the syndrome. One important aspect that arises from this study is the correlation between the adherence to Mediterranean diet and the low risk of frailty in ageing, as many studies confirmed. As in our cases, their difficulty was to assess the correlation of frailty with poor dietary intake by randomized clinical trials. In fact, to date, it seems difficult to find randomized clinical trials that correlate nutritional supplementation and frailty itself. More often, nutritional interventions, based on administration of some micronutrients or proteins, were considered as part of combined intervention in clinical trials or studied in observational studies, therefore no cause-effect relationship was proven.38,39,40,41,42,43,44
Moreover, elderly people experiment the phenomenon called “anorexia of aging”.45,46,47 A significant number of older people fail to ingest sufficient food to meet their essential energy and nutrient needs. In the study of Tsutsumimoto and colleagues, in frail older people the anorexia of aging resulted in 21.2% respect to 7.9% in their robust counterparts, and was independently associated with frailty status, with slowness, exhaustion, and weight loss. Recent systematic reviews seem to confirm these results, such as Kelaiditi et al.,48 that investigated whether nutrition intervention is important to postpone frailty. In contrast, Xu et al.49 performed a systematic review and meta-analysis to assess the ability of protein or amino acid supplementation to augment lean body mass (LBM) or strength of leg muscles in elderly patients, but they did not found any differences in lean body mass gain and muscle strength more than placebo in frail elderly.
As previous said, available evidences are characterized by an overall heterogeneity in terms of objectives and methods, but mostly by the lack of randomized clinical trial performed with certain criteria for frailty definition and inclusion participants. Moreover, the combined interventions found in our revision, did not seem to improve the frailty status more than physical exercise alone, so future study need to be assessed to establish the role of nutrition in the syndrome.
Limits
The most important limit that author encountered in this systematic review, was the scarce prevalence of randomized clinical trials conducted according to univocal criteria for the identification of frailty. Moreover, in most cases trials were conducted including both frail and pre-frail population or institutionalized older adults. The strictly inclusion criteria chosen by the authors were mandatory for the study, but this probably has reduced the number of included studies, reducing the evidence strength. Another important limit was the heterogeneity of interventions, in terms of type, duration and population. These limits, despite a massive literature present in the scientific database, have reduced the quality of randomized clinical trials until now, resulting in the few number of articles comprises in this systematic review.
Conclusion
This systematic review showed that physical intervention alone or combined with nutritional supplementation should improve the frailty status. Furthermore, the role of nutritional intervention alone or in combination is still uncertain and recent data suggest that rather than making interventions based on the supplement of some nutrients, it should be more appropriate a global approach based on a healthy diet. Establishing which exercise program could be more effective to contrast frailty is a challenge for the future. In the meanwhile, one of the most important goal is standardize the screening tools to identify community-dwelling frail people who might benefit from prevention strategy or early interventions.
Therefore, an early identification of people at risk of being frail could significantly reduce the adverse outcomes of the syndrome and improve both the performance of healthcare systems and older people quality of life. Moreover, targeting people who are at risk for frailty, could also lead to economic advantages.
In conclusion this systematic review, although including a small number of articles, confirmed the benefits of physical exercise in reducing the frailty status, but more studies are needed to understand which is the best approach to achieve lasting results. More studies are also auspicial to understand the role of nutrition in the frailty syndrome and its treatment. The authors are convinced of the usefulness of this type of intervention, both isolated and in support of physical activity, so we hope that future studies will focus on this topic.
References
- He W, Goodkind D, Kowal P. An Aging World: 2015 International Population Reports. March 2016 P95/16-1
- Rapporto ISTAT: Bilancio Demografico 2017. https://www.istat.it/it/files/2017/06/bilanciodemografico2016_13giugno2017.pdf?title=Bilancio+demografico+nazionale+-+13%2Fgiu%2F2017+-+Testo+integrale.pdf
- Ageing in the Twenty-First Century: A Celebration and a Challenge. Published by the United Nations Population Fund (UNFPA), New York, and HelpAge International, London. ISBN 978-0-89714-981-5
- Liotta G, O'Caoimh R, Gilardi F, et al. Assessment of Frailty in Community-Dwelling Older Adults Residents in the Lazio Region (Italy): A Model to Plan Regional Community-Based Services. Arch Gerontol Geriatr. 2017; 68: 1-7. doi: 10.1016/j.archger.2016.08.004.
- Li BL, Li W, Bi JQ, Meng QG, Fei JF. Predicting Factors Associated with Frailty in Aged Patients with Bone-Arthrosis Pain in the Clinic. Phys Sportsmed. 2016; 44(4): 391-396.
- Mello Ade C, Engstrom EM, Alves LC. Health-Related and Socio-Demographic Factors Associated with Frailty in the Elderly: A Systematic Literature Review. Cad Saude Publica. 2014; 30(6): 1143-68.
- Abellan van Kan G, Rolland Y, Houles M, Gillette-Guyonnet S, Soto M, Vellas B. The Assessment of Frailty in Older Adults. Clin Geriatr Med. 2010; 26(2): 275-86. doi: 10.1016/j.cger.2010.02.002.
- Fried LP, Tangen CM, Walston J, et al. Cardiovascular Health Study Collaborative Research Group. Frailty in Older Adults: Evidence for a Phenotype. J Gerontol A Biol Sci Med Sci. 2001; 56(3): M146-56.
- Rockwood K, Song X, MacKnight C, et al. A Global Clinical Measure of Fitness and Frailty in Elderly People. CMAJ. 2005; 173(5): 489-95.
- Rockwood K, Stadnyk K, MacKnight C, McDowell I, Hébert R, Hogan DB. A Brief Clinical Instrument to Classify Frailty in Elderly People. Lancet. 1999 16; 353(9148): 205-6.
- Gobbens RJ, Luijkx KG, Wijnen-Sponselee MT, Schols JM. In Search of an Integral Conceptual Definition of Frailty: Opinions of Experts. J Am Med Dir Assoc. 2010; 11(5): 338-43. doi: 10.1016/j.jamda.2009.09.015.
- Chen X, Mao G, Leng SX. Frailty Syndrome: An Overview. Clin Interv Aging. 2014 19; 9:433-41. doi: 10.2147/CIA.S45300
- Walston J, Hadley EC, Ferrucci L, et al. Research Agenda for Frailty in Older Adults: Toward a Better Understanding of Physiology and Etiology: Summary from the American Geriatrics Society/ National Institute on Aging Research Conference on Frailty in Older Adults. J Am Geriatr Soc. 2006; 54(6): 991-1001.
- Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in Elderly People. Lancet. 2013 2; 381(9868):752-62. doi: 10.1016/S0140-6736(12)62167-9.
- Legramante JM, Morciano L, Lucaroni F et al. Frequent Use of Emergency Departments by the Elderly Population when Continuing Care Is Not Well Established. PLoS One. 2016 Dec 14; 11(12):e0165939. doi: 10.1371/journal.pone.0165939.
- Collard RM, Boter H, Schoevers RA, Oude Voshaar RC. Prevalence of Frailty in Community-Dwelling Older Persons: A Systematic Review. J Am Geriatr Soc. 2012 Aug; 60(8):1487-92. doi: 10.1111/j.1532-5415.2012.04054.x.
- Ferrucci L, Guralnik JM, Studenski S, Fried LP, Cutler GB Jr, Walston JD; Interventions on Frailty Working Group. Designing Randomized, Controlled Trials Aimed at Preventing or Delaying Functional Decline and Disability in Frail, Older Persons: A Consensus Report. J Am Geriatr Soc. 2004 Apr; 52(4):625-34.
- Bergman H, Ferrucci L, Guralnik J et al. Frailty: An Emerging Research and Clinical Paradigm – Issues and Controversies. J Gerontol A Biol Sci Med Sci. 2007 Jul; 62(7):731-7.
- Kelaiditi E, van Kan GA, Cesari M. Frailty: Role of Nutrition and Exercise. Curr Opin Clin Nutr Metab Care. 2014 Jan;17(1):32-9. doi: 10.1097/MCO.0000000000000008.
- Liberati A, Altman DG, Tetzlaff J et al. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies that Evaluate Healthcare Interventions: Explanation and Elaboration. BMJ. 2009 Jul 21; 339:b2700. doi: 10.1136/bmj.b2700.
- Langlois F, Vu TT, Chassé K, Dupuis G, Kergoat MJ, Bherer L. Benefits of Physical Exercise Training on Cognition and Quality of Life in Frail Older Adults. J Gerontol B Psychol Sci Soc Sci. 2013 May; 68(3):400-4. doi: 10.1093/geronb/gbs069.
- Schulz KF, Altman DG, Moher D; CONSORT Group. CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomised Trials. BMC Med. 2010 Mar 24; 8:18. doi: 10.1186/1741-7015-8-18.
- Moher D, Hopewell S, Schulz KF et al. CONSORT. CONSORT 2010 Explanation and Elaboration: Updated Guidelines for Reporting Parallel Group Randomised Trials. Int J Surg. 2012; 10(1):28-55. doi: 10.1016/j.ijsu.2011.10.001.
- Giné-Garriga M, Guerra M, Pagès E, Manini TM, Jiménez R, Unnithan VB. The Effect of Functional Circuit Training on Physical Frailty in Frail Older Adults: A Randomized Controlled Trial. J Aging Phys Act. 2010 Oct; 18(4):401-24.
- Tarazona-Santabalbina FJ, Gómez-Cabrera MC, Pérez-Ros P et al. A Multicomponent Exercise Intervention that Reverses Frailty and Improves Cognition, Emotion, and Social Networking in the Community-Dwelling Frail Elderly: A Randomized Clinical Trial. J Am Med Dir Assoc. 2016 May 1; 17(5):426-33. doi: 10.1016/j.jamda.2016.01.019.
- Cameron ID, Fairhall N, Langron C et al. A Multifactorial Interdisciplinary Intervention Reduces Frailty in Older People: Randomized Trial. BMC Med. 2013 Mar 11; 11:65. doi: 10.1186/1741-7015-11-65.
- Zhang L, Weng C, Liu M, Wang Q, Liu L, He Y. Effect of Whole-Body Vibration Exercise on Mobility, Balance Ability and General Health Status in Frail Elderly Patients: A Pilot Randomized Controlled Trial. Clin Rehabil. 2014 Jan; 28(1):59-68. doi: 10.1177/0269215513492162.
- Ng TP, Feng L, Nyunt MS et al. Nutritional, Physical, Cognitive, and Combination Interventions and Frailty Reversal among Older Adults: A Randomized Controlled Trial. Am J Med. 2015 Nov; 128(11):1225-1236.e1. doi: 10.1016/j.amjmed.2015.06.017.
- Chan DC, Tsou HH, Yang RS et al. A Pilot Randomized Controlled Trial to Improve Geriatric Frailty. BMC Geriatr. 2012 Sep 25; 12:58. doi: 10.1186/1471-2318-12-58.
- Cesari M, Vellas B, Hsu FC et al. A Physical Activity Intervention to Treat the Frailty Syndrome in Older Persons-Results from the LIFE-P Study. J Gerontol A Biol Sci Med Sci. 2015 Feb; 70(2):216-22. doi: 10.1093/gerona/glu099.
- Puts MT, Toubasi S, Andrew MK et al. Interventions to Prevent or Reduce the Level of Frailty in Community-Dwelling Older Adults: A Scoping Review of the Literature and International Policies. Age Ageing. 2017 Jan 6. doi: 10.1093/ageing/afw247.
- Dedeyne L, Deschodt M, Verschueren S, Tournoy J, Gielen E. Effects of Multi-Domain Interventions in (Pre)Frail Elderly on Frailty, Functional, and Cognitive Status: A Systematic Review. Clin Interv Aging. 2017 May 24; 12:873-896. doi: 10.2147/CIA.S130794.
- Giné-Garriga M, Roqué-Fíguls M, Coll-Planas L, Sitjà-Rabert M, Salvà A. Physical Exercise Interventions for Improving Performance-Based Measures of Physical Function in Community-Dwelling, Frail Older Adults: A Systematic Review and Meta-Analysis. Arch Phys Med Rehabil. 2014 Apr; 95(4):753-769.e3. doi: 10.1016/j.apmr.2013.11.007.
- Chou CH, Hwang CL, Wu YT. Effect of Exercise on Physical Function, Daily Living Activities, and Quality of Life in the Frail Older Adults: A Meta-Analysis. Arch Phys Med Rehabil. 2012 Feb; 93(2):237-44. doi: 10.1016/j.apmr.2011.08.042.
- de Labra C, Guimaraes-Pinheiro C, Maseda A, Lorenzo T, Millán-Calenti JC. Effects of Physical Exercise Interventions in Frail Older Adults: A Systematic Review of Randomized Controlled Trials. BMC Geriatr. 2015 Dec 2; 15:154. doi: 10.1186/s12877-015-0155-4.
- Daniels R, van Rossum E, de Witte L, Kempen GI, van den Heuvel W. Interventions to Prevent Disability in Frail Community-Dwelling Elderly: A Systematic Review. BMC Health Serv Res. 2008 Dec 30; 8:278. doi: 10.1186/1472-6963-8-278.
- Yannakoulia M, Ntanasi E, Anastasiou CA, Scarmeas N. Frailty and Nutrition: from Epidemiological and Clinical Evidence to Potential Mechanisms. Metabolism. 2017 Mar; 68:64-76. doi: 10.1016/j.metabol.2016.12.005.
- León-Muñoz LM, Guallar-Castillón P, López-García E, Rodríguez-Artalejo F. Mediterranean Diet and Risk of Frailty in Community-Dwelling Older Adults. J Am Med Dir Assoc. 2014 Dec; 15(12):899-903. doi: 10.1016/j.jamda.2014.06.013.
- Rahi B, Ajana S, Tabue-Teguo M, Dartigues JF, Peres K, Feart C. High Adherence to a Mediterranean Diet and Lower Risk of Frailty among French Older Adults Community-Dwellers: Results from the Three-City-Bordeaux Study. Clin Nutr. 2017 May 31. pii: S0261-5614(17)30194-2. doi: 10.1016/j.clnu.2017.05.020.
- Kelaiditi E, Jennings A, Steves CJ et al. Measurements of Skeletal Muscle Mass and Power Are Positively Related to a Mediterranean Dietary Pattern in Women. Osteoporos Int. 2016 Nov; 27(11):3251-3260. doi: 10.1007/s00198-016-3665-9.
- Chan R, Leung J, Woo J. Dietary Patterns and Risk of Frailty in Chinese Community-Dwelling Older People in Hong Kong: A Prospective Cohort Study. Nutrients. 2015 Aug 24; 7(8):7070-84. doi: 10.3390/nu7085326.
- Talegawkar SA, Bandinelli S, Bandeen-Roche K et al. A Higher Adherence to a Mediterranean-Style Diet Is Inversely Associated with the Development of Frailty in Community-Dwelling Elderly Men and Women. J Nutr. 2012 Dec; 142(12):2161-6. doi: 10.3945/jn.112.165498.
- Bollwein J, Diekmann R, Kaiser MJ et al. Dietary Quality Is Related to Frailty in Community-Dwelling Older Adults. J Gerontol A Biol Sci Med Sci. 2013 Apr; 68(4):483-9. doi: 10.1093/gerona/gls204.
- Urpi-Sarda M, Andres-Lacueva C, Rabassa M et al. The Relationship Between Urinary Total Polyphenols and the Frailty Phenotype in a Community-Dwelling Older Population: The InCHIANTI Study. J Gerontol A Biol Sci Med Sci. 2015 Sep; 70(9):1141-7. doi: 10.1093/gerona/glv026.
- Tsutsumimoto K, Doi T, Makizako H et al. The Association Between Anorexia of Aging and Physical Frailty: Results from the National Center for Geriatrics and Gerontology's Study of Geriatric Syndromes. Maturitas. 2017 Mar; 97:32-37. doi: 10.1016/j.maturitas.2016.12.005.
- Malafarina V, Uriz-Otano F, Gil-Guerrero L, Iniesta R. The Anorexia of Ageing: Physiopathology, Prevalence, Associated Comorbidity and Mortality. A Systematic Review. Maturitas. 2013 Apr; 74(4):293-302. doi: 10.1016/j.maturitas.2013.01.016.
- Di Francesco V, Fantin F, Omizzolo F et al. The Anorexia of Aging. Dig Dis. 2007; 25(2):129-37.
- Kelaiditi E, Guyonnet S, Cesari M. Is Nutrition Important to Postpone Frailty? Curr Opin Clin Nutr Metab Care. 2015 Jan; 18(1):37-42. doi: 10.1097/MCO.0000000000000129.
- Xu ZR, Tan ZJ, Zhang Q, Gui QF, Yang YM. Clinical Effectiveness of Protein and Amino Acid Supplementation on Building Muscle Mass in Elderly People: A Meta-Analysis. PLoS One. 2014 Sep 30; 9(9):e109141.