The impact of malaria on child growth: anthropometric outcomes in a pediatric HIV-exposed cohort in Malawi
Scarcella P,* Moramarco S,* Buonomo E,* Nielsen-Saines K,§ Jere H,@ Guidotti G,° Palombi L,* Marazzi MC#
*Department of Biomedicine and Prevention, University of Rome Tor Vergata
#LUMSA University, Rome
@DREAM Program, Community of Sant’Egidio, Malawi
§UCLA, USA
°ASL ROMA 1, Rome
Introduction
Prevention of HIV mother-to-child transmission is a multidimensional challenge for Sub-Saharan African countries, particularly in the case of Malawi. The DREAM program (Community of Sant’Egidio) provides cART Option B+ to HIV-infected mothers since 2012, with breastfeeding promotion until 12 months of age and dietary supplementation with local foods in order to support the growth and health of children.
Materials and Methods
A cohort of 1586 HIV-exposed uninfected Malawian children (age 0-18 months) were followed from January 2012 to December 2013. Breastfeeding was exclusive for the first 6 months, then complementary feeding was initiated with breastfeeding up to 12 months. Children were monitored for growth, HIV/AIDS, malnutrition, malaria and other health problems. Weight-for-age, length-for-age, weight-for-length z-scores (WAZ, LAZ, WLZ) were calculated with ANTHRO-WHO software. Statistical analysis was computed using the SPSS software (v.20).
Results
In regards to the assessment of child growth, mean values of WAZ (1 m: -0.68 ±1.2; 3 m.: -0.65 ±1.21; 6 m: -0.76 ±1.22; 12 m: -0.94 ±1.12; 18 m: -1.03 ±1.02), LAZ (1 m: -1.30 ±1.33; 3 m: -1.4 ±1.32; 6 m: -1.34 ±1.18; 12 m: -1.5 ±1.12; 18 m: -1.7 ±1.08), and WLZ (1 m: 0.59 ±1.32; 3 m: 0.75 ±1.36; 6 m: 0.21 ±1.26; 12 m: -0.25 ±1.11; 18 m: -0.3 ±1.02), were comparable to previous results reported for similar cohorts. A progressive decline in values for anthropometric measurements was noted over time, with a negative peak at 18 months of age, shortly after the interruption of breastfeeding.
A binary logistic regression was performed in order to better understand the impact of illness on growth faltering. Compared to the other infections (diarrhea, fever, general respiratory infections), malaria was the only significant predictor of general malnutrition at 18 months of age (chronic malnutrition/LAZ<-2 HR: 1.3, p=0.05; underweight/WAZ<-2 HR: 1.4, p=0.04; acute malnutrition/WLZ<-2 HR: 2.1, p=0.005). The association between malaria and poor nutritional status became even stronger when considering indicators of severe malnutrition (LAZ<-3 HR: 1.5, p=0.02; WAZ<-3 HR: 2.4, p=0.005), with the highest association found between malaria and WLZ <-3 (HR: 5.7, p=0.007). Children who had malaria were at greater risk of being severely malnourished at 18 months of age, especially when considering indicators of acute malnutrition (wasting OR: 5.5, CI 1.2-24.9; underweight OR: 2.8, CI 1.3-5.9).
Conclusions
Maternal breastfeeding remained a robust protective factor associated with improved growth and health in HIV-exposed uninfected children. Among pediatric infections malaria stands out as a significant deterrent to adequate nutritional development in HIV-exposed African children. Our findings underlie the negative temporal association between malaria and nutritional status, while highlighting the vicious cycle of malnutrition and malaria.
INTRODUCTION
The advances in mother-to-child prevention of HIV transmission (PMTCT) programs leading to the increased use of effective combination antiretrovirals (cART) has resulted in large numbers of HIV exposed uninfected (HEU) children in many settings. There is substantial evidence that women with HIV are more likely to have infants who are small for gestational age1,2 and that HIV-exposed infants are more likely to be preterm or low birth weight.3 Moreover, children born to HIV-positive women are particularly susceptible to malnutrition,4 while malnutrition during childhood can interfere with the development of acquired protective immune responses to infections.5,6,7
Malnourished children who do not quickly break away from the vicious cycle of infectious diseases and growth failure are vulnerable to irreversible cognitive damage.8 HEU children are at higher risk of morbidity9 and mortality10,11 than their non-exposed peers, even because their mothers become ill or die and cannot provide usual care.12 A recent study demonstrated that acute and chronic malnutrition decrease the odds of attaining adequate motor milestones even among HEU children.13 These findings highlight the need for early nutritional monitoring, counseling and support for HEU children,14 especially in low-income setting where growth failure is partially attributable to inadequate complementary feeding.15
The 2013 WHO Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection maintain that the primary aim of HIV and infant feeding recommendations is to improve HIV-free survival by reducing the risk of transmission through breastmilk using ARVs, while avoiding malnutrition and the increased risk of serious infections in infants and children from unsafe feeding practices.16 The guidelines recommended that mothers with HIV in low resource/high-HIV burden settings receive the same infant and young child feeding (IYCF) advices as HIV-uninfected mothers: early initiation of breastfeeding within 1 hour of birth, exclusive breastfeeding (EBF) for the first 6 months, followed by introduction of appropriate complementary foods at 6 months with continued breastfeeding.17 WHO guidelines support the benefits of both breastfeeding and that of ARVs on mother-infant pairs, emphasizing the significant reduction in postnatal HIV transmission through this approach. Moreover, continued breastfeeding for the first 12 months of life protects infants from mortality, diarrhea, pneumonia and malnutrition, thanks to the high levels of nutrients in breast milk. The 2013 revision of WHO guidelines for HIV PMTCT,18 which recommends lifelong antiretroviral treatment to all HIV-infected women coupled with continuing breastfeeding until 12 months of age, has been endorsed by a growing number of countries.19 These recommendations have favorably impacted Malawian children, with reported increments in growth parameters20,21 and reduced rates of HIV MTCT in the country.22
MATERIALS AND METHODS
Data was pooled from all Drug Resource Enhancement against AIDS and Malnutrition (DREAM) centers in Malawi for a retrospective cohort study. All data were extracted from the DREAM database. DREAM is a public health program aimed at the prevention and treatment of HIV/AIDS with multiple centres in 10 African countries. It was designed and managed by the Community of Sant’Egidio, an Italian faith-based non-governmental organization. The DREAM Program’s approach to the diagnosis, treatment, prevention of PMTCT and growth monitoring of HIV exposed children in Africa has been described in prior studies.23,24 The study was approved by the National Health Sciences Research Committee of Malawi (approval number NHSRC #16/6/1615).
Study Population, Setting and Data Collection
The study population included HIV-exposed children born between January 2012 and December 2013 to HIV positive women attending the DREAM Program antenatal clinics in Malawi. All mothers came from low socioeconomic households and received antiretroviral therapy and monthly nutritional supplementation as part of program guidelines.
Following the 2013 revision of the WHO guidelines for the prevention on mother-to-child transmission of HIV (PMTCT), the DREAM program introduced Option B+ in order to maintain safe breastfeeding.25 Breastfeeding for up to 12 months was supported through health education and food supplementation.
The majority of pregnant women, irrespective of CD4 levels, started tenofovir, lamivudine, and efavirenz in pregnancy and continued indefinitely after delivery, with a 12 month recommended duration of breastfeeding. Infants received nevirapine for 6 weeks.
Health monitoring included medical history, birth weight/weight in the first week of life, general laboratory parameters (hemoglobin and transaminases), and HIV-1 infection status measured by HIV DNA PCR at 1 month of age and HIV-1 bDNA assays at 6, and 12 months of age. Children were followed for all medical problems as well as diagnosis of HIV/AIDS infection. Parents (or guardians) were encouraged to bring their children to the clinic whenever they were ill. Specific medications were provided for the treatment of illnesses. Malaria was diagnosed via microscopy examination of thick and thin blood smear and positive cases were treated with artemisinin. Malaria cases were defined based on clinical and laboratory findings.
Anthropometric and nutritional assessments
Anthropometric parameters were monthly measured and registered through a software package obtained in collaboration with the Department of Nutrition of the World Health Organization.26,27 In accordance with international standards, periodic anthropometric assessments included monthly measurement of weight and length. Growth monitoring was performed until 18 months. Children were measured without clothing or footwear; weight (in kilograms) was determined using a mechanical baby scale (SECA 745—Class III), while length (in centimeters) was assessed by laying children on their back and using a stadiometer for newborn babies and infants (SECA 210). Staff involved in assessing measurements were appropriately trained.
Anthropometric indicators were calculated using Anthro WHO software. Nutritional status was assessed using the following standardized anthropometric z-scores as per WHO 2006 standards:28 length-for-age z- score (LAZ) for stunting, weight-for-length z-scores (WLZ) for wasting and weight-for-age z-score (WAZ) for underweight. Following WHO criteria, general malnutrition was defined as: stunting/LAZ <-2, wasting/WLZ <-2, and underweight/WAZ <-2. Moreover, severe malnutrition was defined as LAZ <-3 (severe stunting), WLZ <-3 (severe wasting), WAZ <-3 (severe underweight). Malnutrition was assessed and managed in accordance with the Malawian guidelines for the management of malnutrition.29 Prevalence of malnutrition (stunting, wasting, underweight) was evaluated at 6 and 18 months of age in children categorized into two groups according to malaria event: children who did have malaria (CHM) and children who did not have malaria (CHNM).
Statistical Analysis
Pediatric records were extracted from the DREAM database. Data were analyzed using SPSS software system 20.0 (IBM Somers, NY, USA). Descriptive data and variables measured were presented as means with standard deviations (SD). WHO Anthro Software (Version 3.2.2, January 2011) was used to compute children’s weight-for-age (WAZ), weight-for-length (WLZ), and length-for-age (LAZ) z-scores per WHO standards. Differences between means of anthropometric parameters and z-score of malnutrition indices between CHM and CHNM groups were tested with the Student’s t-test. Univariate (OR CI 95%) and multivariate logistic regressions (HR CI 95%) were performed in order to identify predictors of general and severe malnutrition among the clinical illnesses (fever, diarrhea, general respiratory infections, malaria) at 18 months. At the same age, the Odds Ratios, 95% confidence intervals, between malaria and severe malnutrition indices were calculated.
RESULTS
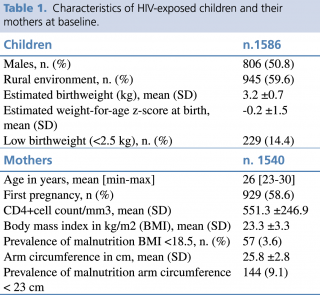
Data for a total of 1586 HIV-exposed uninfected children (51% males) born during the study period were included in the analysis. All children were breastfed until at least 12 months of age (with exclusive breastfeeding for the first 6 months). Baseline maternal data and characteristics of the children are reported in Table 1
Table 1. Characteristics of HIV-exposed children and their mothers at baseline
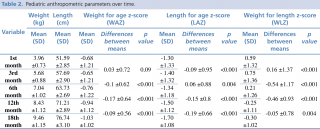
For the pediatric growth assessment, Table 2 reports the mean Z-scores for anthropometric measurements and parameters over time: mean values of weight (1m: 3.96 ±0.73; 3 m: 5.68 ±0.88; 6 m: 7.04 ±1.02; 12m: 8.43 ±1.12; 18 m:9.46 ±1.15); length (1m: 51.59 ±2.85; 3m: 57.69 ±2.90; 6 m:63.73 ±2.69; 12m: 71.21 ±2.89; 18m: 76.74 ±3.10); WAZ (1 m: -0.68 ±1.21; 3 m: -0.66 ±1.21; 6 m: -0.76 ±1.22; 12 m: -0.94 ±1.12; 18 m: -1.03 ±1.02), LAZ (1 m: -1.30 ±1.33; 3 m: -1.40 ±1.32; 6 m: -1.34 ±1.18; 12 m: -1.50 ±1.12; 18 m: -1.70 ±1.08), WLZ (1 m: 0.59 ±1.32; 3 m: 0.75 ±1.36; 6 m: 0.21 ±1.26; 12 m: -0.25 ±1.11; 18 m: -0.30 ±1.02) were comparable to previous results conducted in similar cohorts.30 The differences between mean z-scores were statistically significant. A progressive decline in anthropometric measurements was noted over time, with a negative peak at 18 months of age, coinciding with the period immediately following interruption of breastfeeding.
Table 2. Pediatric anthropometric parameters over time
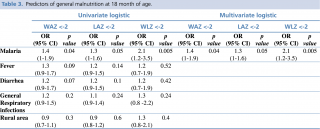
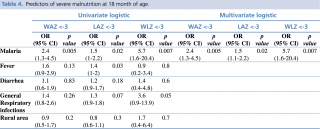
For the purposes of statistical analysis children at 18 months of age were divided into distinct groups: malnourished children versus not malnourished children; children who experienced specific clinical events (such as fever, diarrhea, malaria, general respiratory infection) as reported on electronic records versus children who did not have any such events. Comparisons were also made between children residing in rural versus urban environments. Univariate logistic regression was used to investigate whether clinical events were associated with chronic and acute malnutrition (stunting, underweight and wasting); odds ratios were used to quantify the association between each risk factor and poor growth. Multivariate analyses were then computed in order to better understand the impact of illness on growth faltering and to define the main predictor of malnutrition. Compared to the other infections (diarrhea, fever, general respiratory infection), malaria was the only significant predictor of general malnutrition (z-scores <-2) at 18 months of age for all anthropometric indicators (Table 3). The impact of malaria became significantly stronger when considering severe malnutrition indicators (Table 4) (LAZ<-3 HR: 1.5 p=0.02; WAZ<-3: HR: 2.4 p=0.005), with the highest risk reported for acute malnutrition (WLZ <-3 HR: 5.7 p=0.007). Residence in a rural environment was not associated with increased risk of malnutrition.
Table 3. Predictors of general malnutrition at 18 month of age
Table 4. Predictors of severe malnutrition at 18 month of age
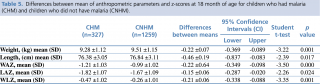
To better understand the impact of malaria on nutritional status, differences between mean Z-scores for anthropometric indicators were investigated, comparing children who experienced malaria with children who did not. At 18 months of age, differences between means for all anthropometric parameters and indices were significantly lower in children affected by malaria (weight: -0.22 ±0.07, p=0.001; length: -0.46 ±0.19, p=0.017; WAZ: -0.22 ±0.64, p<0.001; LAZ: -0.15 ±0.06, p=0.024; WLZ: -0.21 ±0.06, p=0.001).
Table 5. Differences between mean of anthropometric parameters and z-scores at 18 month of age for children who had malaria (CHM) and children who did not have malaria (CNHM)
Among 327 children who had malaria we selected a subgroup of 170 children for whom anthropometric parameters and relatives z-scores were available before and after the episode of malaria (mean month of malaria event 10.2 ±2.5 SD; min 6.07 – max 14.9).
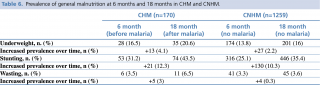
General malnutrition (defined as stunting/chronic malnutrition: LAZ <-2 SD; wasting/acute malnutrition: WLZ<-2 SD; underweight: WAZ <-2 SD) was evaluated both before and after the first episode of malaria. Although the prevalence of malnourished children generally increased over time in both groups, Table 6 shows that the prevalence of malnourishment in children who had malaria consistently increased at 18 months (after the malaria episode) and was higher than that of children who had not experienced a malaria episode (underweight: 20.6% vs 16%; stunting: 43.5% vs 35.4%; wasting: 6.5% vs 3.6%). Conversely, children with poor nutritional status at 6 months could potentially be more vulnerable to malaria infection. In fact, at 6 months the prevalence of malnutrition in the malaria group of children was higher than in the non-malaria counterpart even before the first episode of malaria, especially for underweight and stunting. The prevalence of wasting was similar in both groups initially but increased by over 3% in children who had experienced malaria at the 18 months of age assessment.
Table 6. Prevalence of general malnutrition at 6 months and 18 months in CHM and CNHM
Results of our study underscore the negative association between malaria infection and nutritional status. A strong association between malaria and subsequent malnutrition was noted, with a higher and significant association with acute malnutrition noted, a finding that had not been previously demonstrated in studies to date. On the other hand, our study did not find a statistically significant association between malaria and stunting potentially because our cohort included younger aged children who were not included in other studies reporting such an association.31
The presence of a temporal association between malaria and malnutrition in our cohort, leads us to conclude that malaria induced growth faltering. Nevertheless, we also noted that most children were already suffering from general malnutrition before their first episode of malaria. It appears that malaria contributed to the worsening nutritional status of affected children, leading to severe acute malnutrition. As a vicious cycle developed, severe acute malnutrition potentially impaired the immune system, thus contributing to further episodes of malaria.32
CONCLUSION
Maternal breastfeeding is a well-documented promoter of growth and health maintenance in HIV-exposed pediatric populations. Within the spectrum of early pediatric infections, malaria stands out as a significant deterrent to adequate nutritional development.
Our findings emphasize the importance of prevention and treatment of malnutrition in HIV-exposed children associated with an effective control of malaria infection.
Acknowledgments
We would like to acknowledge with gratitude the Community of Sant’Egidio, all the staff and the participating patients of the Malawian DREAM centres for their contribution to this study.
References
1 Taha TE, Dallabetta GA, Canner JK, Chiphangwi JD, Liomba G, Hoover DR, et al.: The Effect of Human Immunodeficiency Virus Infection on Birthweight and Infant and Child Mortality in Urban Malawi. Int J Epidemiol. 1995; 24:1022–1029.
2 Ndirangu J, Newell ML, Bland RM, Thorne C: Maternal HIV Infection Associated with Small-Forgestational Age Infants but Not Preterm Births: Evidence from Rural South Africa. Hum Reprod.2012; 27:1846–1856.
3 Arpadi SM: Growth Failure in HIV-Infected Children. WHO Consultation on Nutrition and HIV/AIDS in Africa: Evidence, Lessons and Recommendations for Action, Durban, South Africa, 10–13 April 2005. Geneva: WHO; 2005
4 Arpadi S, Fawzy A, Aldrovandi GM, Kankasa C, Sinkala M, Mwiya M, et al.: Growth Faltering Due to Breastfeeding Cessation in Uninfected Children Born to HIV-Infected Mothers in Zambia. Am. J. Clin. Nutr. 2009; 90(2):344-53. doi: 10.3945/ajcn.2009.27745.
5 Gaayeb L, Sarr JB, Cames C, Pinçon C, Hanon JB, Ndiath MO, et al.: Effects of Malnutrition on Children's Immunity to Bacterial Antigens in Northern Senegal. Am J Trop Med Hyg. 2014 Mar; 90(3):566-73. doi: 10.4269/ajtmh.12-0657.
6 Hoang T, Agger EM, Cassidy JP, Christensen JP, Andersen P: Protein Energy Malnutrition during Vaccination Has Limited Influence on Vaccine Efficacy but Abolishes Immunity if Administered during Mycobacterium Tuberculosis Infection. Infect Immun. 2015 May; 83(5):2118-26. doi: 10.1128/IAI.03030-14.
7 Ozkan H, Olgun N, Saşmaz E, Abacioğlu H, Okuyan M, Cevik N: Nutrition, Immunity and Infections: T Lymphocyte Subpopulations in Protein—Energy Malnutrition. J Trop Pediatr. 1993 Aug; 39(4):257-60
8 Victora CG, Adair L, Fall C, Hallal PC, Martorell R, Richter L, et al.: Maternal and Child Undernutrition: Consequences for Adult Health and Human Capital. Lancet 2008, 371, 340–357.
9 Filteau S: The HIV-Exposed, Uninfected African Child. Trop Med Int Health. 2009; 14:276–287.
10 Marinda E, Humphrey JH, Iliff PJ, Mutasa K, Nathoo KJ, Piwoz EG, et al.: Child Mortality according to Maternal and Infant HIV Status in Zimbabwe. Pediatr. Infect Dis J. 2007; 26:519–526.
11 Landes M, van Lettow M, Chan AK, Mayuni I, Schouten EJ, Bedell RA: Mortality and Health Outcomes of HIV Exposed and Unexposed Children in a PMTCT Cohort in Malawi. PloS One. 2012; 7: 1–7.
12 Palombi L, Marazzi MC, Voetberg A, Magid NA: Treatment Acceleration Program and the Experience of the DREAM Program in Prevention of Mother-To-Child Transmission of HIV. AIDS 2007; 21, Suppl 4: S65-71.
13 Buonomo E, Scarcella P, Tembo D, Giuliano M, Moramarco S, Palombi L, et al.: Malnutrition Decreases the Odds of Attaining Motor Milestones in HIV Exposed Children: Results from a Pediatric DREAM Cohort. Epidemiol. Prev. 2015, 39, 108–112.
14 Owor M, Mwatha A, Guay LA: Long Term Follow-up of Children in the HIVNET 012 Perinatal HIV Prevention Trial: Five-Year Growth and Survival. J Acquir Immune Defic Syndr. 2013; 64(5): 464–471. doi:10.1097/QAI.0000000000000015.
15 Victora CG, de Onis M, Hallal PC, Blössner M, Shrimpton R: Worldwide Timing of Growth Faltering: Revisiting Implications for Interventions. Pediatrics 2010, 125, e473-e480.
16 World Health Organization (WHO): Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection: Recommendations for a Public Health Approach. Geneva: WHO; 2013.
17 WHO, UNICEF, UNAIDS, UNFPA. Guidelines on HIV and Infant Feeding: Principles and Recommendations for Infant Feeding in the Context of HIV and a Summary of Evidence. Geneva: WHO; 2010.
18 World Health Organization (WHO): Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection. Recommendations for a Public Health Approach. Geneva, Switzerland: World Health Organization, 2013
19 O'Brien L, Shaffer N, Sangrujee N, Abimbola TO: The Incremental Cost of Switching from Option B+ for the Incremental Cost of Switching from Option B to Option B+ for the Prevention of Mother-To-Child Transmission of HIV. Bull World Health Organ. 2014; 92: 162-170. doi:10.2471/BLT.13.122523.
20 Kamudoni P, Maleta K, Shi Z, Holmboe-Ottesen G: Exclusive Breastfeeding Duration during the First 6 Months of Life Is Positively Associated with Length-For-Age among Infants 6-12 Months Old, in Mangochi District, Malawi. Eur J Clin Nutr. 2015 Jan; 69(1):96-101.
21 Palombi L, Pirillo MF, Andreotti M, Liotta G, Erba F, Sagno JB, al.: Antiretroviral Prophylaxis for Breastfeeding Transmission in Malawi: Drug Concentrations, Virological Efficacy And Safety. Antivir Ther. 2012; 17(8):1511-9. doi: 10.3851/IMP2315.
22 Marazzi MC; Liotta G; Nielsen-Saines K; Haswell J; Abdul Magid N; Buonomo E; Scarcella P; Doro Altan AM; Mancinelli S; Palombi L. Extended Antenatal Antiretroviral Use Correlates with Improved Infant Outcomes Throughout the First Year of Life AIDS 2010, 24: 2819-2826
23 Liotta G, Floridia M, Andreotti M, Jere H, Sagno JB, Marazzi MC, et al.: Growth Indices in Breastfed Infants Pre and Postnatally Exposed to Tenofovir Compared with Tenofovir-Unexposed Infants. AIDS. 2016 Jan 28;30(3):525-7. doi: 10.1097/QAD.0000000000000944.
24 Giuliano M, Andreotti M, Liotta G, Jere H, Sagno JB, Maulidi M, et al.: Maternal Antiretroviral Therapy for the Prevention of Mother-To-Child Transmission of HIV in Malawi: Maternal and Infant Outcomes Two Years After Delivery. PLoSOne. 2013 Jul 19; 8(7):e68950. doi: 10.1371/journal.pone.0068950. Print 2013.
25 World Health Organization (WHO): Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing HIV Infection Recommendations for a Public Health Approach. Geneva, Switzerland 2013
26 Nucita A, Bernava GM, Bartolo M, Di Pane Masi F, Giglio P, Peroni M et al.: A Global Approach to the Management of EMR (Electronic Medical Records) of Patients with HIV/AIDS in Sub-Saharan Africa: the Experience of DREAM Software. BMC Med Inform Decis Mak 2009; 9; doi: 10.1186/1472-6947-9-42.
27 World Health Organization (WHO): Anthro for Personal Computers, Version 3.1. Software for Assessing Growth and Development of The World’s Children. Geneva (Switzerland), WHO, 2010. http://www.who.int/childgrowth/software/en/ (accessed January12, 2012).
28 World Health Organization (WHO): Multicentre Growth Reference Study Group: WHO Child Growth Standards: Methods and Development. Geneva, Switzerland: World Health Organization; 2006.
29 Guidelines for the Management of Moderate Acute Malnutrition. Blantyre (Malawi), Government of Malawi, Ministry of Health, 2006.
30 Liotta G, Floridia M, Andreotti M, Jere H, Sagno JB, Marazzi MC, et al.: Growth Indices in Breastfed Infants Pre and Postnatally Exposed to Tenofovir Compared with Tenofovir-Unexposed Infants. AIDS. 2016 Jan 28; 30(3):525-7. doi: 10.1097/QAD.0000000000000944.
31 Ferreira EA, Alexandre MA, Salinas JL, de Siqueira AM, Benzecry SG, de Lacerda MVG, Monteiro WM: Association between Anthropometry-Based Nutritional Status and Malaria: A Systematic Review of Observational Studies. Malar J. 2015 Sep 17; 14:346. doi: 10.1186/s12936-015-0870-5
32 Arinaitwe E, Gasasira A, Verret W, Homsy J, Wanzira H, Kakuru A, et al.: The Association Between Malnutrition and the Incidence of Malaria among Young HIV-Infected and Uninfected Ugandan Children: A Prospective Study. Malar J. 2012 Mar 27; 11:90. doi: 10.1186/1475-2875-11-90.