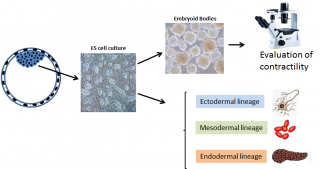
The multiple functions of pluripotent cells: can Embryoid Bodies be a screening tool for environmental toxicity?
Luisa Campagnolo, Micol Massimiani, Giuseppina Somma, Andrea Magrini and Antonio Pietroiusti
Department of Biomedicine & Prevention, School of Medicine and Surgery, University of Rome “Tor Vergata”, Rome, Italy
Embryonic Stem Cells and Their Differentiation
The development of embryonic stem (ES) cell lines at the beginning of the 80’s has represented one of the biggest scientific revolutions of the last century. These lines were originally derived from the inner cell mass of the mouse blastocysts, which in vivo contributes to the formation of the three germ layers (ectoderm, mesoderm and endoderm) and all their derivatives.1,2 The isolation and in vitro culture of cells of the inner cell mass has allowed not only to elucidate most of the molecular pathways governing stemness, but also to generate transgenic animals to study gene function. After being initially developed from mice, ES cell lines have been derived from the blastocyst of many other mammalian species, including humans.3 Under appropriate culture conditions, ES cells can be differentiated into tridimensional structures called embryoid bodies (EBs), which recapitulate in vitro embryonic development, by differentiating derivatives of all three germ layers. Indeed, ability to form EBs is considered a pre-requisite to classify a newly developed cell line as a bona fide ES cell line (Figure 1). During EB differentiation, the spatiotemporal expression of early genes within the three germ layers follows that observed in vivo, demonstrating similarities between the two differentiation programs.4 Indeed, by using a panel of early expressed genes or lineage markers (such as for example oct-3/4, Brachyury T and GATA-4 or flk-1, Nkx-2.5 and Msx3), it was observed that the progressive differentiation of EB well correlated with the early development of the post-implantation mouse embryo, thus demonstrating that EB may be exploited for studies of lineage determination in mammals. One of the peculiarities of the EB is the spontaneous differentiation of contracting cardiomyocyte-like cells, which can be easily microscopically visualized. This feature has been exploited to develop rapid and cost effective in vitro screening systems to screen chemical compounds for their embryotoxic potential.
Figure 1. Graphical representation of the potential use of mES. Embryonic stem cells can be differentiated in vitro to give embryoid bodies in which the development of contracting cardiomyocyte-cells can be microscopically monitored. Under specific culture conditions the differentiation of several cell lineages can be obtained.
The Embryonic Stem Cell Test
After the proposal of the “three Rs” principle over 50 years ago, the need to reduce the use of animal testing for toxicological purposes has stimulated the development of alternative screening protocols. Refinement of in vitro methods for screening the toxicity of different compounds has become a whole field of research and at the European levels a Committee for the validation of these alternative methods has been established (European Committee for the Validation for Alternative Methods, ECVAM). Differentiation of embryonic stem cells into contracting EB has gained great interest, as disturbances by different chemicals and drugs of this program could be easily visualized under the microscope and interpreted as a sign of embryotoxicy. The complementary interaction between biologists and computational mathematicians, has allowed to design a new test called the Embryonic Stem cell Test (EST).5 In the EST, beside the mouse ES cells (to represent the embryonic tissue), a second cell line, the embryonic fibroblast NIH3T3 cell line (to represent the differentiated tissue) is used. By stepwise discriminant analysis, three endpoints were selected as best representing the correlation with the embryotoxic properties of test chemicals, which are the doses inhibiting by 50% the proliferation of the two cell lines (IC50) and the dose inhibiting by 50% the differentiation of contracting cardiomyocytes in the EB (ID50). These three values are then inserted in an algorithm that allows classifying a specific substance as strong, weak or non embryotoxic. The use of this in vitro test has been endorsed by the ECVAM in 2002 and listed as an alternative to in vivo prenatal developmental toxicity studies (OECD Test Guideline 414). In the original protocol, differentiation of EB is obtained by the “hanging drop” system. Basically, 20 ml drops of medium containing 800 cells are seeded on the lid of a tissue culture plate. After 3 days, the drops are transferred in a petri dish filled with differentiation medium and allowed to grow for additional two days, after which each single EB is transferred in the well of a 24 well-plate and cultured for 5 more days, allowing the differentiation of contracting cardiomyocytes. Since its validation, several modifications have been proposed to improve test duration and limit operator dependent manipulation and decisions. Recently, to limit size heterogeneity of the EB, the use of 96 well-plates with V-shaped bottom has been suggested.6 In addition, introduction of an automated image recording system to evaluate the area of contractility (in pixels) and the frequency of contraction (in Hz) has been proposed as an unbiased analysis of embryotoxic potency of the test compound.7 A further improvement toward a high throughput screening test introduces the use of fluorescence activated cell sorting (FACS) analysis, through which the differential expression of molecular markers of heart differentiation can be studied. The FACS-EST presents two advantages: i. the expression of marker proteins (such as sarcomeric myosin heavy chain and alpha-actinin) can be unequivocally evaluated, reducing the contribution of operator dependent experience and judgement; ii. the duration of the test can be reduced to 7 days, as the onset of protein expression precedes contractility.8 More recently, in an attempt to improve translation of the EST to humans, the use of human stem cell lines to differentiate EB has been proposed.9
Applications of the EST
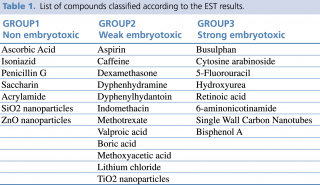
During the validation procedure, 20 different chemical substances have been classified in one of the three EST categories and the in vitro results compared to in vivo data. So far the EST has been used by pharmaceutical and cosmetic companies for drug testing. Tables with the classification of several compounds as strong weak and non-embryotoxic are available (for a summary see table 1).
Table 1. List of compounds classified according to the EST results
Over the last five years, we and other groups have demonstrated that the EST can be exploited to predict the embryotoxic potential of engineered nanoparticles (NPs). Thanks to peculiar properties that make them very different from their micrometric counterpart, NPs are intentionally produced particles in the nanometric range (1-100 nm) which find application in many industrial and biomedical products. As a consequence of their wide use, criticisms have been raised toward their safety for human health, especially for vulnerable populations, such as pregnant women and the unborn child. By using the EST, we have classified a specific type of carbon based nanoparticles, the single wall carbon nanotubes (SWCNT) as strong embryotoxic, and have demonstrated a robust correlation between in vitro and in vivo data.10 More recently, we have exploited the EST to classify metal based nanoparticles containing silica, zinc and titanium (SiO2NP, ZnONP and TiO2NP), which differently from the SWCNT, fell into the non-embryotoxic (SiO2NP and ZnONP) or weak embryotoxic group (TiO2NP).11 Also for these NPs, the prediction of the EST was confirmed by in vivo results (Campagnolo, unpublished data). More recently, differentiation of contracting cardiomyocyte-like cells in the EB has been successfully used to evaluate toxicity of the environmental pollutants, such as several pesticides and Bisphenol A,12 proving the concept that the EST might be exploited to assess toxicity of many other pollutants without the costs of time-consuming animal models.
Conclusion and Open Questions
The EST represents a valuable in vitro screening test to predict embryotoxic potential of different compounds with different chemical and physical characteristics. The positive correlation between the EST results and the parallel data obtained in vivo for some environmental pollutants and engineered nanoparticles, which resembles the ultrafine particulate matter, clearly points toward the possible wide use of the test for the screening of environmental pollutants. The many proposed modifications to the original protocol can definitely improve the reliability of the test, and might be considered in the development of a more sophisticated high throughput screening platform to evaluate the embryotoxic potential of environmental contaminants. One main point needs to be further investigated and refined: per se, the EST can be considered as a conservative test, meaning that it gives information on the possible embryotoxic potential of the tested substance directly put in contact with the simulated embryo. Nevertheless, the tested substance could fail to reach in vivo the developing embryo, due to the presence of the placenta, which may function as a non-permeable barrier for some substances. Thus the classification obtained by the EST might be more severe than the reality. Although precautionary results are safer especially in regulatory contests, they might impair the use of pregnancy-safe products or could wrongly classify substances present in the environment as teratogens. In this respect, the development of a more sophisticated test which considers the use of a simulated placental barrier is desired.
References
1. Evans MJ, Kaufman MH. Establishment in Culture of Pluripotential Cells from Mouse Embryos. Nature. 1981 Jul 9; 292(5819):154-6.
2. Martin GR. Isolation of a Pluripotent Cell Line from Early Mouse Embryos Cultured in Medium Conditioned by Teratocarcinoma Stem Cells. Proc Natl Acad Sci U S A. 1981 Dec; 78(12):7634-8.
3. Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. Embryonic Stem Cell Lines Derived from Human Blastocysts. Science. 1998 Nov 6; 282(5391):1145-7.
4. Leahy A, Xiong JW, Kuhnert F, Stuhlmann H. Use of Developmental Marker Genes to Define Temporal and Spatial Patterns of Differentiation During Embryoid Body Formation. J Exp Zool. 1999 Jun 15; 284(1):67-81.
5. Genschow E, Spielmann H, Scholz G, Pohl I, Seiler A, Clemann N, Bremer S, Becker K. Validation of the Embryonic Stem Cell Test in the International ECVAM Validation Study on Three In Vitro Embryotoxicity Tests. Altern Lab Anim. 2004 Sep; 32(3):209-44.
6. Peters AK, Steemans M, Hansen E, Mesens N, Verheyen GR, Vanparys P. Evaluation of the Embryotoxic Potency of Compounds in a Newly Revised High Throughput Embryonic Stem Cell Test. Toxicol Sci. 2008 Oct;105(2):342-50.
7. Peters AK, Wouwer GV, Weyn B, Verheyen GR, Vanparys P, Gompel JV. Automated Analysis of Contractility in the Embryonic Stem Cell Test, A Novel Approach to Assess Embryotoxicity. Toxicol In Vitro 2008 Dec;22(8):1948-56
8. Buesen R, Genschow E, Slawik B, Visan A, Spielmann H, Luch A, Seiler A. Embryonic Stem Cell Test Remastered: Comparison Between the Validated EST and the New Molecular FACS-EST for Assessing Developmental Toxicity In Vitro. Toxicol Sci. 2009 Apr; 108(2):389-400.
9. Hong EJ, Jeung EB. Assessment of Developmental Toxicants Using Human Embryonic Stem Cells. Toxicol Res. 2013 Dec 31; 29(4):221-7.
10. Farcal L, Torres Andón F, Di Cristo L, Rotoli BM, Bussolati O, Bergamaschi E, Mech A, Hartmann NB, Rasmussen K, Riego-Sintes J, Ponti J, Kinsner-Ovaskainen A, Rossi F, Oomen A, Bos P, Chen R, Bai R, Chen C, Rocks L, Fulton N, Ross B, Hutchison G, Tran L, Mues S, Ossig R, Schnekenburger J, Campagnolo L, Vecchione L, Pietroiusti A, Fadeel B. Comprehensive In Vitro Toxicity Testing of a Panel of Representative Oxide Nanomaterials: First Steps towards an Intelligent Testing Strategy. PLoS One. 2015 May 21; 10(5):e0127174.
11. Pietroiusti A, Massimiani M, Fenoglio I, Colonna M, Valentini F, Palleschi G, Camaioni A, Magrini A, Siracusa G, Bergamaschi A, Sgambato A, Campagnolo L. Low Doses of Pristine and Oxidized Single-Wall Carbon Nanotubes Affect Mammalian Embryonic Development. ACS Nano. 2011 Jun 28;5(6):4624-33.
12. Yin N, Yao X, Qin Z, Wang YL, Faiola F. Assessment of Bisphenol A (BPA) Neurotoxicity In Vitro with Mouse Embryonic Stem Cells. J Environ Sci (China). 2015 Oct 1; 36:181-7.